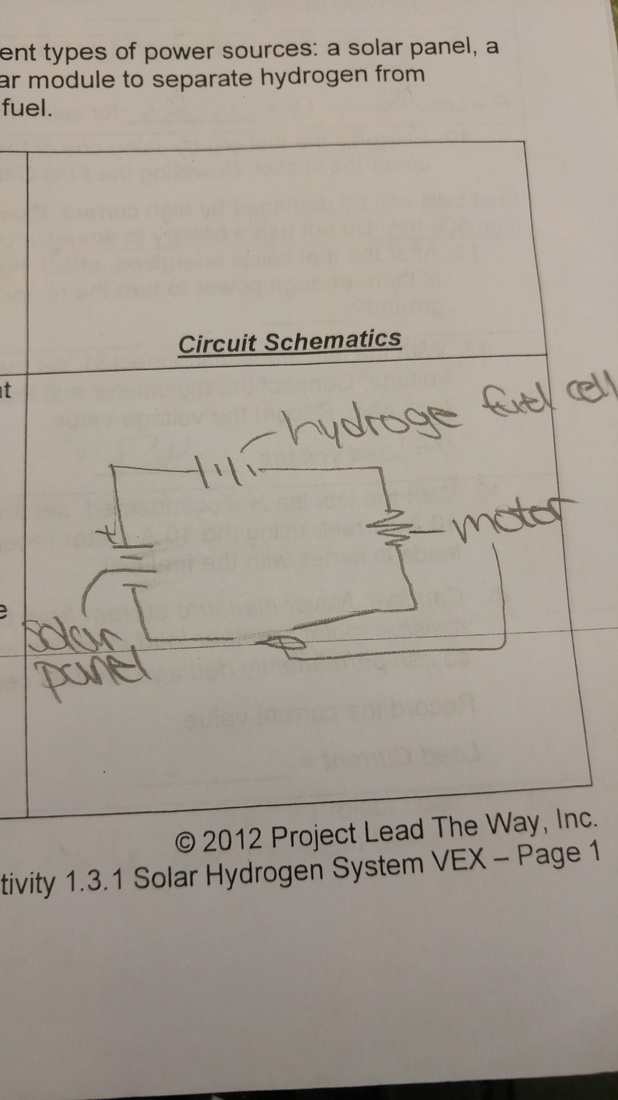
hydrogen fuel cell
A fuel cell is a device that generates electricity by a chemical reaction. Every fuel cell has two electrodes, anode and cathode. The purpose of a fuel cell is to produce an electrical current that can be directed outside the cell to do work. First, hydrogen atoms enter a fuel cell at the anode where a chemical reaction strips them of their electrons. The hydrogen atoms are now "ionized," and carry a positive electrical charge. The negatively charged electrons provide the current through wires to do work.
solar panels
solar panels works by allowing photons, or particles of light, to knock electrons free from atoms, generating a flow of electricity. Solar panels actually comprise many, smaller units called photovoltaic cells. Each cell generates a few volts of electricity and the panel combines the energy they produce to make a bigger electric current and voltage. The cells are made from silicon, a very common chemical element found in sand. When sunlight shines on a solar cell, the energy it carries blasts electrons out of the silicon.
series and parallel circuits
In a series circuit, the current through each of the components is the same, and the voltage across the circuit is the sum of the voltages across each component. In a parallel circuit, the voltage across each of the components is the same, and the total current is the sum of the currents through each component.
circuit
pictorial representation
reflection
I felt this project was complicated and time consuming. When we try to measure the current or resistance, we have to set up the hydrogen fuel cell and wait about 10 minutes. Also, we didn't had time to measure the current and resistance. Built the ca, which was easiest or least I thought took most of our time. Our group didn't know what to do except for we need to built the car and measures. Still, I felt accomplished When our car moved.
activity 1.3.1
Your browser does not support viewing this document. Click here to download the document.
conclusion
1. Using the measurements you made, compare and relate the four options you explored. Was the car best powered by a single fuel cell, a single solar module, two AAA batteries in series, or two AAA batteries in parallel?
In a series circuit, the current through each of the components is the same, and the voltage across the circuit is the sum of the voltages across each component. In a parallel circuit, the voltage across each of the components is the same, and the total current is the sum of the currents through each component.
2. Did voltage, current, or power best describe the suitability of a power source?
No. Because, it won't allow motor to turn constantly.
3. If you had many solar modules, how many of them would be needed to get the same performance from the car as the performance observed with two AAA batteries? Describe or sketch how would you connect the solar modules in terms of parallel and series circuits.
we need 2 solar modules. Because 1 solar module produce 1.5v, so we need 2 solar module to produce 3v.
4. If you had many fuel cells, how many of them would be needed to get the same performance from the car as the performance observed with two AAA batteries? Describe or sketch how would you connect the fuel cells in terms of parallel and series circuits.
5. Describe and defend a system that you believe would best utilize a solar hydrogen system to meet the needs for an average driver.
Put a solar panel on top of a car. During the day, make sure that it gets the most sunlight possible. It will then power the car. Because it is better for the environment. Also, the driver will save money, by not having to pay for gas.
6. How does a photovoltaic cell work? Record the source of your information.
It is like a solar cell panel. It absorbs the light of the sun (or another light source), it takes that and turns it into energy that we can use.
7. Detail how electrolysis separates hydrogen and oxygen. How is electricity produced as the fuel cell allows the hydrogen to reunite in a bond with oxygen? Record the source of your information
Electrolysis separates hydrogen and oxygen because it strips the extra electron off of Hydrogen as it joins the Oxygen atom.